In recent years, nucleic acid drugs have become a key area of global research and development and investment. Compared with traditional drugs that carry out disease intervention at the protein level, nucleic acid drugs can play a role at the source of genetic information transmission, so they have advantages such as strong specificity, rich gene targets and lasting curative effect. Besides, they can significantly reduce the production cost by avoiding the complicated synthesis and purification process of traditional drugs.
Since the double helix structure of DNA was discovered in 1953, nucleic acid drugs have been developed for 70 years. The types include antisense nucleic acid (ASO), small interfering RNA (siRNA), micrornas (mirnas), small activating RNA (saRNA), messenger RNA (mRNA), aptamer, ribozyme, antibody nucleic acid coupling drug (ARC), etc. In addition to mRNA drugs, basically other nucleic acid drugs are composed of less than 100 ribonucleotides or deoxyribonucleotides single or double chains, so they are also called oligonucleotides or small nucleic acid drugs.
Different from mRNA drugs that encode proteins, oligonucleotide drugs pair with DNA, mRNA or pre-mRNA mainly through the principle of base complementary pairing, and regulate gene expression through a series of mechanisms such as gene silencing, non-coding RNA inhibition and gene activation.
At present, there are 17 nucleic acid drugs on the market in the world, of which 3 have applied for the market, and 32, 115 and 113 types of clinical pipeline in phase III, Phase II and Phase I are under research respectively. Among the 719 nucleic acid drugs in the world, ASO accounted for the largest number (253), followed by siRNA(229) and mRNA(72). Ionis (91 products), Alnylam(34 products), simaomics(25 products), arrowhead (24 products) and Astrazeneca (18 products) are the TOP5 companies in the global investment layout of nucleic acid drugs.
Since most nucleic acid drugs are oligonucleotides, oligonucleotides become an important raw material of nucleic acid drugs, and oligonucleotides are formed by the series of dozens of nucleotides, so nucleoside monomer is also a key raw material of nucleic acid drugs.
According to the forecast of QYR, the sales volume of global nucleotide market (including oligonucleotides and monomers) will reach 510 million US dollars in 2021. China is the largest nucleotide production market in the world, accounting for about 55%. The current domestic nucleic acid drug raw material market is estimated to be 280 million US dollars, in addition to the global nucleotide market demand is about 50,000 tons, and the annual output value of the domestic nucleotide market is about 27,500 tons. Most of them are exported, and the domestic internal digestion capacity for nucleic acid drug production is weak. This is not unrelated to the comparison between nucleic acid drugs production and foreign countries.

Because overall, 90% of the world’s nucleotides are used for nucleic acid drugs, and only about 10% are used for health food, agriculture and other fields.
From the perspective of oligonucleotide raw material production technology, the current mainstream method of oligonucleotide synthesis in the upstream process is solid phase phosphophosphate triester method, in which the phosphophosphate monomer and solid phase carrier through the steps of deprotection, coupling, oxidation, capping, etc., to obtain the target base sequence, that is, oligonucleotide crude. After removing the protective group, the crude product was cut off from the solid phase carrier. After the purity of the crude product was preliminarily detected by liquid chromatography, the intermediates of oligonucleotides were obtained through purification, ultrafiltration, lyophilization and other steps.
Taking DNA primer synthesis as an example, the following figure shows the steps of solid phase phosphite synthesis. In the purification process, In industry, there are mainly reverse phase chromatography (solid phase synthesis – reverse phase chromatography purification – detriphenylmethylation – ethanol precipitation, etc.), ion exchange chromatography (solid phase synthesis – ion exchange chromatography – concentration/deep filtration – freeze drying), hydrophobic chromatography combined with ion exchange chromatography (solid phase synthesis – hydrophobic chromatography – detriphenylmethylation – ion exchange – UF/DF – bulk drug solution ), but most nucleic acid drugs on the market are prepared by the first two methods. In addition to the chemical synthesis methods mentioned above, other methods for the preparation of nucleotides include RNA enzymatic hydrolysis, microbial fermentation and biocatalyzation.
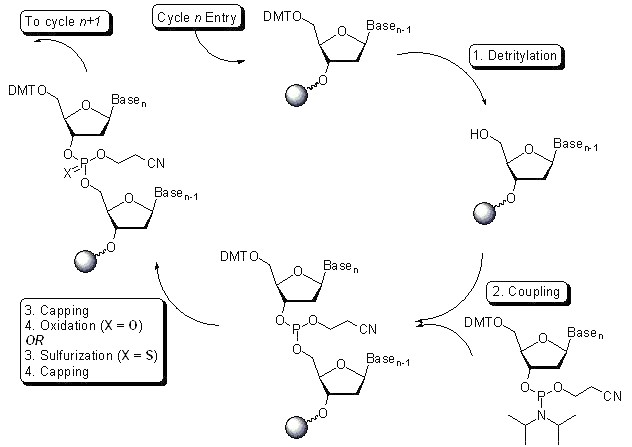
Oligonucleotides and nucleoside monomers are the key materials of nucleic acid apis, and qualified suppliers that meet the quality requirements are relatively rare in the world. This is mainly due to the solid phase synthesis technology used in the production of nucleic acid apis, which has high barriers in process development, process amplification and quality control. Besides, the initial investment in supporting equipment and clean environment for solid phase synthesis of nucleic acid apis is very large, and the production must meet the requirements of international GMP.
The few international TOP10 qualified suppliers are Danaher Corporation (founded in 1969), Merck KGaA (founded in 1668), Eurofins Scientific SE (founded in 1987) and Thermo Fisher Scientific Inc. (founded in 2006), Agilent Technologies, Inc. (f. 1999), GE Healthcare (f. 1994), Kaneka Eurogentec S.A. (f. 1985), GeneDesign, Inc. (f. 2000), LGC Biosearch Technologies (f. 1993), Bio-synthesis Inc. (f. 1984)
There are about 160 suppliers of nucleotide apis that can be queried in China, with dispersed regional distribution, including 43 enterprises in Shaanxi, 22 in Jiangsu, 21 in Hubei, 21 in Hubei, 20 in Shandong, 16 in Guangdong, 11 in Anhui, 6 in Zhejiang, 6 in Shanghai, 5 in Henan, 5 in Hunan, 3 in Tianjin and 2 in Shanxi.
Representatives of domestic oligonucleotide manufacturers are Guangzhou Ruibo, Hequan Pharmaceutical, Shanghai Zhaowei, Yunbio, etc.
From the global point of view, the overall supply of nucleotide raw materials is in short supply and still belongs to the global shortage of products. With the gradual release of clinical potential of nucleic acid drugs in the future, it is bound to drive the continuous growth of related raw materials in the future, and the future prospect is good. As a domestic enterprise, it should focus on the technological research and development of innovative nucleic acid drugs, increase the capacity of self-production and self-marketing, and grow into a leading nucleic acid drug enterprise on par with international giants as soon as possible.



